News
Articles & Recipes
Articles
News
Articles & Recipes
Articles
Articles
An Introduction to pH, pH Adjusting and Formulating with Cosmetic Acids
pH is one of the technical terms you come across frequently in the cosmetic industry. A simple way to think about it is to read it as an indication of how acidic or alkaline your product is, typically on a scale of 1-15. In a cosmetic sense, both ends of the spectrum are classified as being harsh and potentially damaging to skin and as such, cosmetic formulators work to avoid creating products that sit at these extremes.
In technical terms pH is a measure of the number of hydrogen ions in an aqueous solution so, for a product to have a measurable pH there has to be water around for the hydrogen ions to swim around in.
Don't Get Water In Your Salt Scrub
A common way of assessing a cosmetic ingredient is to explore what it mixes with and for many, that starts with whether the ingredient is oil or water soluble. The fact that salt is both readily soluble in water and insoluble in oil is used to our advantage when making salt scrubs, allowing us to harness their exfoliant properties in a spreadable emollient base. The aesthetic of a salt scrub can vary from dry and granular, viscous paste-like solids through to those with a flowable oily top layer blanketing a crystalline salty bottom. Unlike in many cosmetic formulations, scrubs which lack homogeneity and that form these layers are not damaged or broken or less attractive to the public. Indeed, salt scrubs are enduringly popular in all their guises, thanks to their simplicity, wholesomeness and the way they leave your skin feeling.
When Changing Emulsifier Changes Everything
An experiment investigating what happens inside a cream when you change the emulsifier.
Your choice of emulsifier impacts much more than just how stable your cream will be. The right emulsification strategy will improve your product efficacy - how well it does what you say it will do. The emulsifier also plays an important role in the products rheology (how it flows), how it looks and feels and how it ages over time.
Experimenting With Different Natural Preservatives In A Cream Base
Many of our clients struggle when it comes to choosing a preservative for their cream, milk or lotion. In fact, it's such a source of anxiety for some they choose to avoid water-containing formulations altogether- a strategy that can sometimes work but at other times be very limiting.
The Curious State of Glyceryl Stearate
Until last week this commonly-used cosmetic chemical had been something I'd never, ever thought of looking into. I hadn't questioned its chemistry or function having used it many times in many different formulations and while I knew of and appreciated the difference between the SE and non-SE versions of this (SE = Self Emulsifying indicating the presence of a little saponified fatty acid to help turn up its emulsification properties) beyond that, I had next to no interest in spending time analysing this. That has since changed.
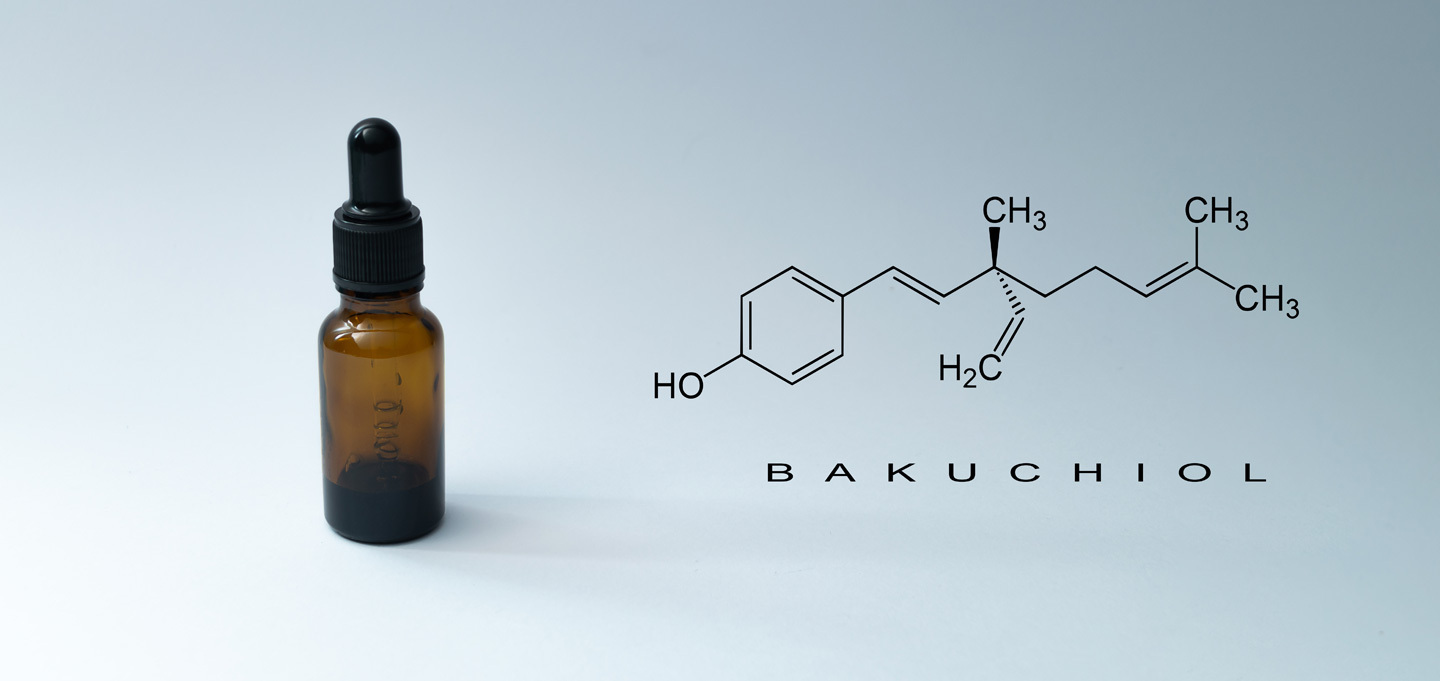
Investigating Bakuchiol - Is It Really Nature’s Retinol?
Around the world, every day of the year there are scientists running experiments. Most of these trials come and go without so much of a mention, becoming just another small drop in an ocean of knowledge. But every now and then an experiment produces a result that becomes a story, a story that becomes an opportunity and an opportunity that becomes something tangible to the masses. It feels like this is the case for Bakuchiol, at least under the circumstances it currently finds itself under. It feels like this is Bakuchiol's skincare moment and to find out why, we're going to read this book backwards. Starting with the final chapter.
Pop A Little Mushroom in Your Skincare
What words come to your mind when you first think about adding mushroom extract to your face creams? Whatever your initial reaction there is a banquet of skin-friendly chemistry to be had inside each grain of mushroom powder and many safe and interesting ways to harness them. Further, it's highly likely you've already benefited from fungi chemistry in many unforeseen ways!
Protein, The Stuff of Hair
While it is true that both the skin and hair are protein rich materials, protein science really comes into its own once we start formulating hair care products.
The structural and functional integrity of hair can be attributed to a protein called Keratin. There are many types of keratin in the natural world ranging from that which you find on hooved animals and tortoise shells through to the softer keratins of feathers, fur and even to the sticky, gloopy mess that is the slime from an eel-like creature called a hagfish! Human hair keratin is most similar to sheep's wool although it is unwise to consider them to be interchangeable as while they look very much the same on initial analysis, dig a bit deeper and you find some very different properties not least the way they deal with hygral stress (water logging).
The Science of Suspension, Transforming A Gel Cleanser Into An Exfoliating Wash
Adding exfoliant particulates to your surfactant-based formulations is a great way to wow your customers and deliver better product performance at the same time. With so many naturally derived, attractive exfoliant materials available it is no wonder that everyone is trying to achieve the perfect gel-based scrub! But you cannot simply add exfoliants to a surfactant base and expect them to stay put. Gravity quickly takes its toll leaving you a hot mess that is practically unsaleable. So how do we overcome that?
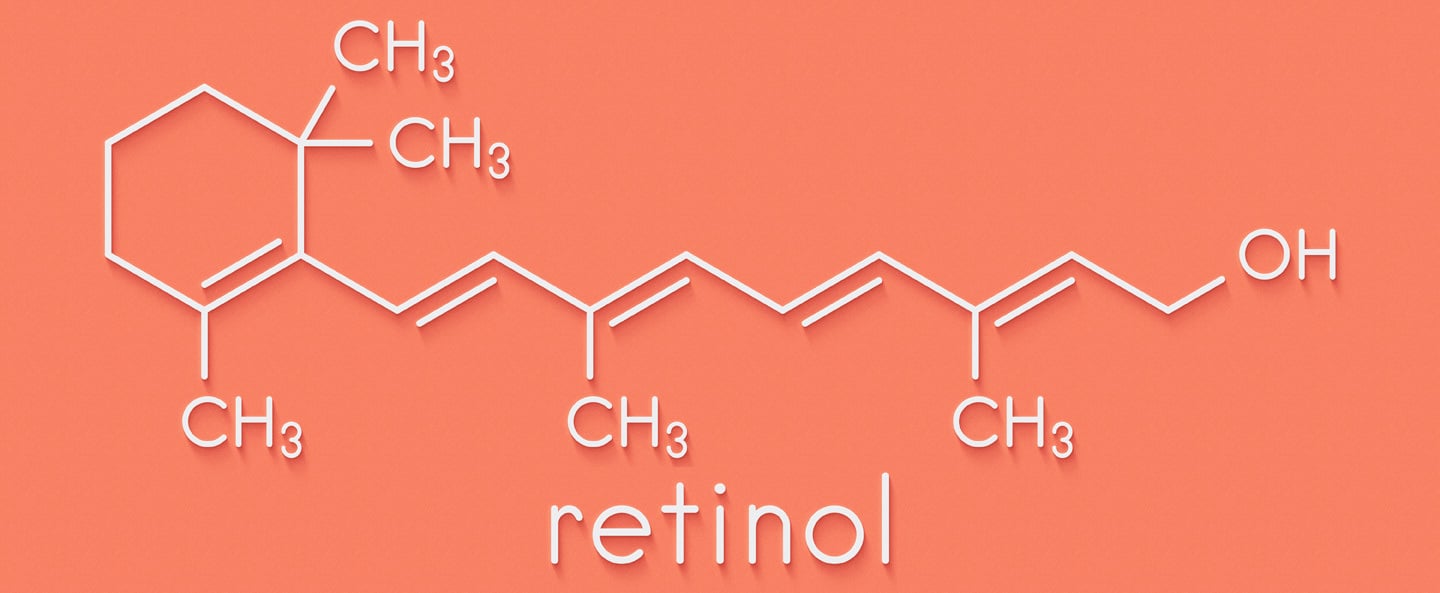
Vitamin A Chemistry
It's difficult to get far in the anti-aging skincare world without coming across or at least considering vitamin A. Vitamin A receptor proteins exist within our skin ready to capture and employ it for the production, differentiation and normalisation of skin cells (keratinocytes), doing this by communicating with keratin genes. Vitamin A not only grows skin, it normalises, even optimises the process, facilitating production of natural moisturising fluid and suppressing excess oil secretions. As a Consequence of topically applied vitamin A is able to suppress, resolve and prevent the formation of pimples, increased skin hydration, decrease fine lines and wrinkle and improve barrier functioning- all highly desirable outcomes that deliver visible aesthetic improvements (1).